Психиатрия Психиатрия и психофармакотерапия им. П.Б. Ганнушкина
K.R. Evgrafov Regional mental hospital 440026, Russian Federation, Penza, ul. Lermontova, d. 28. skrembler@mail.ru
Abstract
The discovery of the first antipsychotic drug - chlorpromazine - took place in 1950 and since then the list of antipsychotics has expanded significantly. These drugs are used successfully for psychotic disorders and some other disorders. The article discusses not only the main areas of application of antipsychotic drugs, but also highlights the main clinically significant problems in their use, as well as the prevention and correction of side effects.
Keywords: antipsychotics, antipsychotics in Russia, neuroleptics, neuroleptics in Russia.
For citation: K.A. Maslov. Antipsychotics in Russia, in modern clinical practice of the psychiatrist. Questions of applied practical application. Psychiatry and psychopharmacotherapy. 2020; 5: 41–50.
Согласно международной анатомо-терапевтическо-химической классификации (далее АТХ), антипсихотические препараты относятся к группе препаратов для лечения заболеваний нервной системы N5 (психолептики), подгруппе N05A (антипсихотические препараты) [1].
Ранее (с конца 1950-х годов) при обозначении АП применяли термины: «нейролептики» [2, 3], «нейроплегики», «большие транквилизаторы» (по аналогии с «малыми транквилизаторами»), «атарактики» [2]. В настоящее время в России используют термин «антипсихотики первого поколения» (далее АПП), «типичные нейролептики», «типики», «классические нейролептики»; за рубежом — first generation antipsychotics или FGA. Существуют также препараты, называемые в России антипсихотиками второго поколения (далее АВП), «атипичные нейролептики», «атипики», или «атипичные антипсихотики») [3], а за рубежом — atypical antipsychotics (AAP), second generation antipsychotics (SGA). По мнению ряда исследователей, основные различия между ААП и АВП заключаются в разном рецепторном профиле и, как следствие, в разной степени представленности и выраженности лечебных и побочных эффектов (прежде всего по выраженности экстрапирамидных расстройств) [4].
Первый АП — хлорпромазин — был открыт в 1950 г. [9] в процессе синтеза антигистаминных препаратов [10]. Этот препарат сразу и надолго стал «золотым стандартом» и эталоном среди АП. Затем были синтезирован ряд других АП, среди которых наибольшее применение получил галоперидол (в клинической практике с 1959 г.) [11].
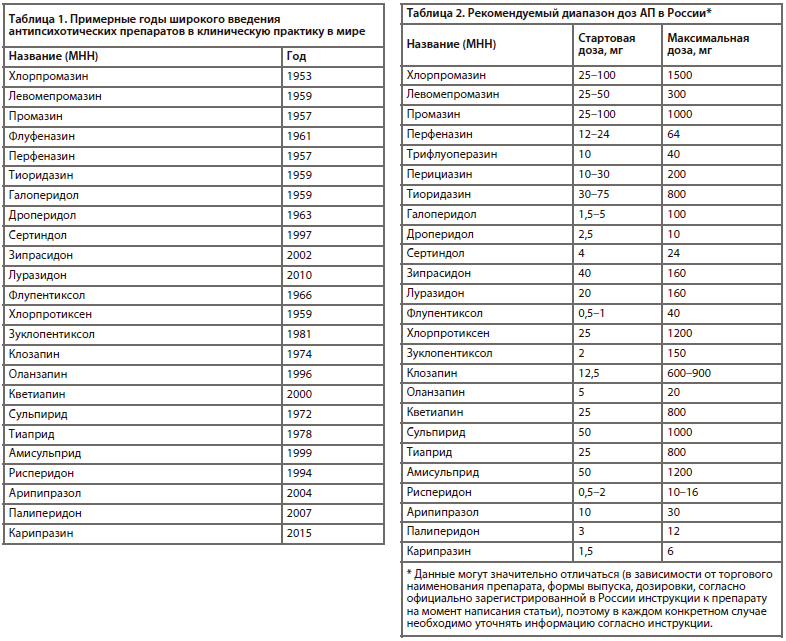
В табл. 1 отражено время широкого внедрения некоторых АП в клиническую практику [10-19].
• антипсихотический эффект (основной);
• седативный эффект;
• нормотимический эффект.
Системное воздействие препарата влечет за собой и ряд побочных эффектов, которые могут вызывать ряд осложнений. В первую очередь, они обусловлены дофаминергической блокадой в мезокортикальном, нигростриальном и тубероинфундибулярном путях, а также влиянием на различные рецепторы в периферических отделах нервной системы. Наиболее часто в числе осложнений встречаются нейролептические экстрапирамидные расстройства (ЭПР), метаболические нарушения (ожирение, диабет, гиперпролактинемия), избыточная седация, вторичная дефицитарная симптоматика, слабость, когнитивные нарушения, нейролептическая депрессия, психозы отмены, сексуальные нарушения, слюнотечение. Отмечается также кардио- и гепатотоксичность АП.
Соотношение выраженности лечебных и побочных эффектов между собой определяет спектр клинического действия препарата.
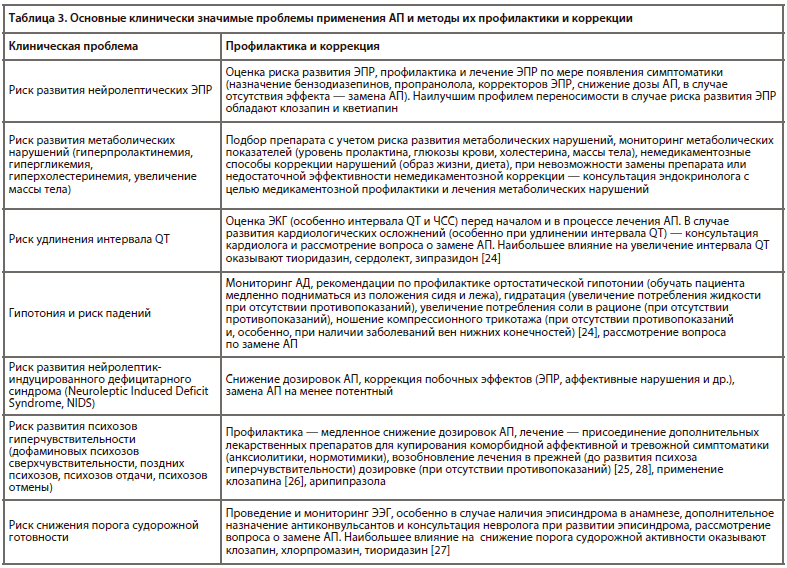
Для большинства АП (кроме, вероятно, клозапина и некоторых современных атипичных АП) развитие антипсихотического эффекта происходит при блокаде, приблизительно, 60–65% дофаминовых рецепторов D2-типа. При блокаде свыше 70–80% рецепторов увеличивается риск развития ЭПР [20-2]. В соответствии с этим условно выделяют «терапевтическое окно» дозировок, в котором АП вызывают собственно антипсихотический эффект. Рекомендуемый диапазон доз АП в России (стартовые и максимальные суточные) [23] представлен в табл. 2.
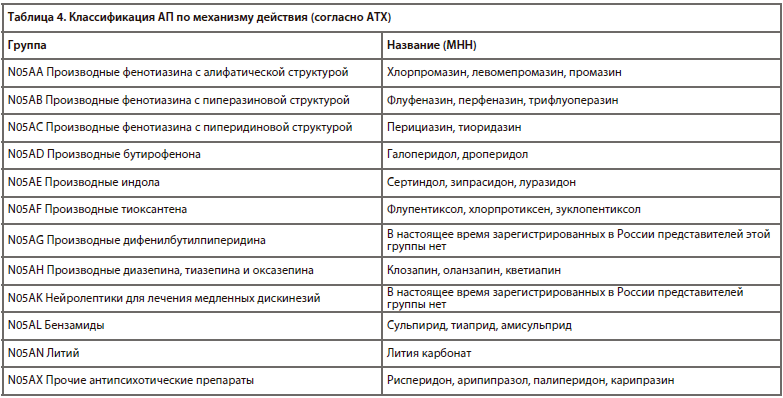
Основные клинически значимые проблемы применения АП и методы их профилактики и коррекции представлены в табл. 3.
В настоящее время в России (по некоторым оценкам) [29-31] наиболее популярны из группы АП в клинической практике (в порядке убывания): рисперидон, сульпирид, тиоридазин, кветиапин.
Для сравнения — в США (по некоторым оценкам) [32] из группы АП наиболее популярны в клинической практике (в порядке убывания): кветиапин, арипипразол, рисперидон.
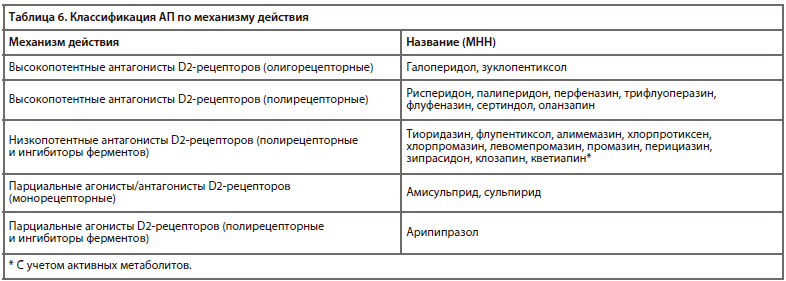
Зарегистрированные в настоящее время в России АП могут подразделяться по их механизму действия, согласно АТХ (табл. 4) [1].
АП также подразделяют в зависимости от поколения на типичные и атипичные (по механизму действия, представленности и выраженности побочных и лечебных эффектов) [4] (табл. 5).
Помимо перечисленных классификаций в отечественной психиатрии существовало, множество различных эклектичных классификаций АП по различным неспецифичным признакам, однако в настоящее время они устарели.
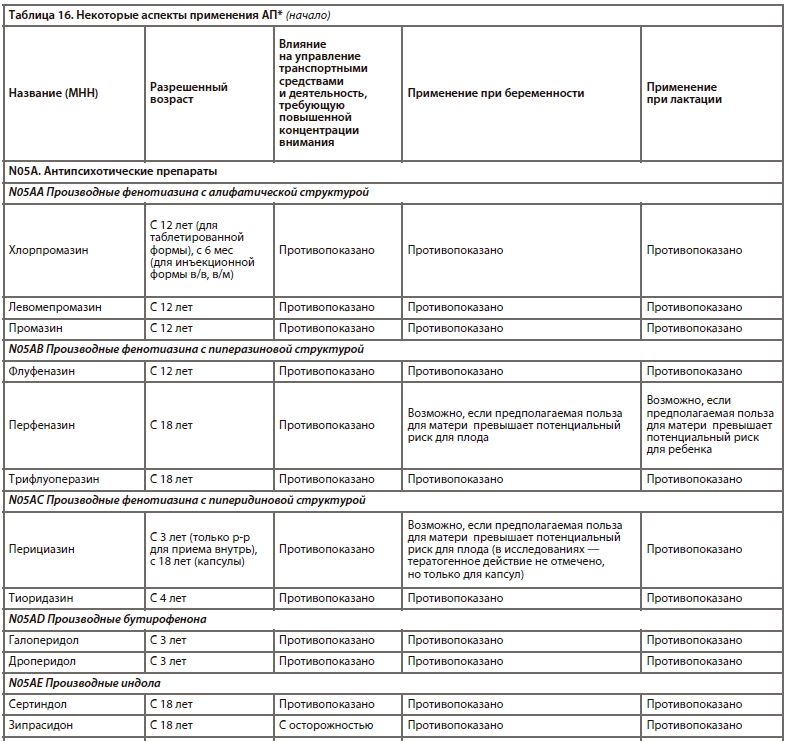
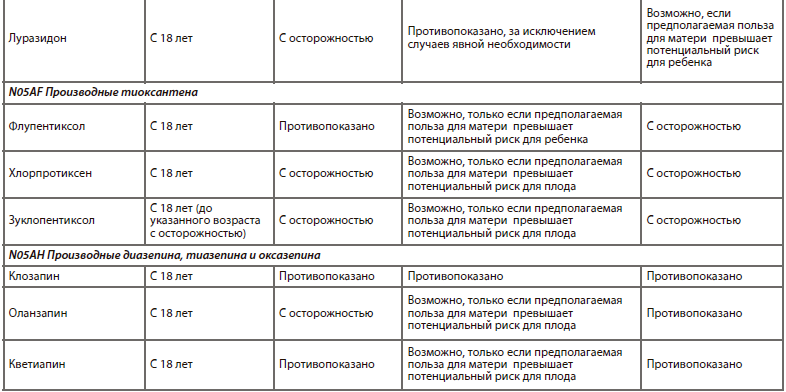
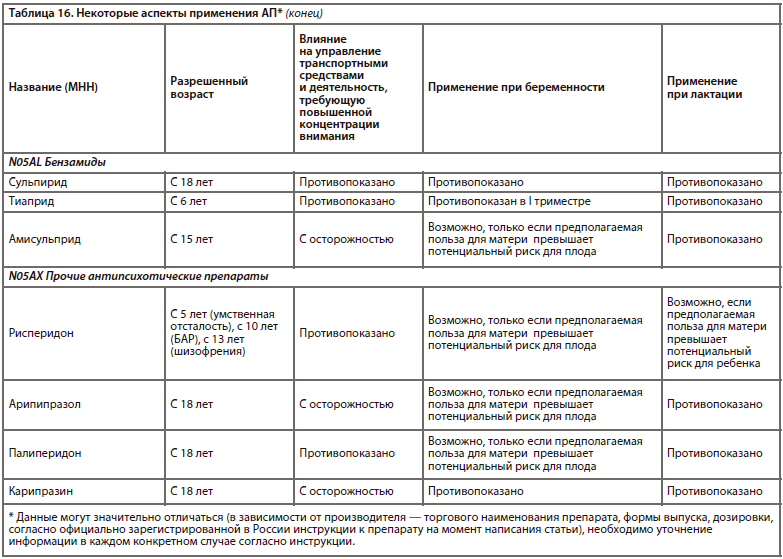
Одна из наиболее прогрессивных и логичных классификаций АП основана на рецепторном механизме действия [26]. Такой подход логичен, последователен и отлично дополняет существующие классификации (табл. 6).
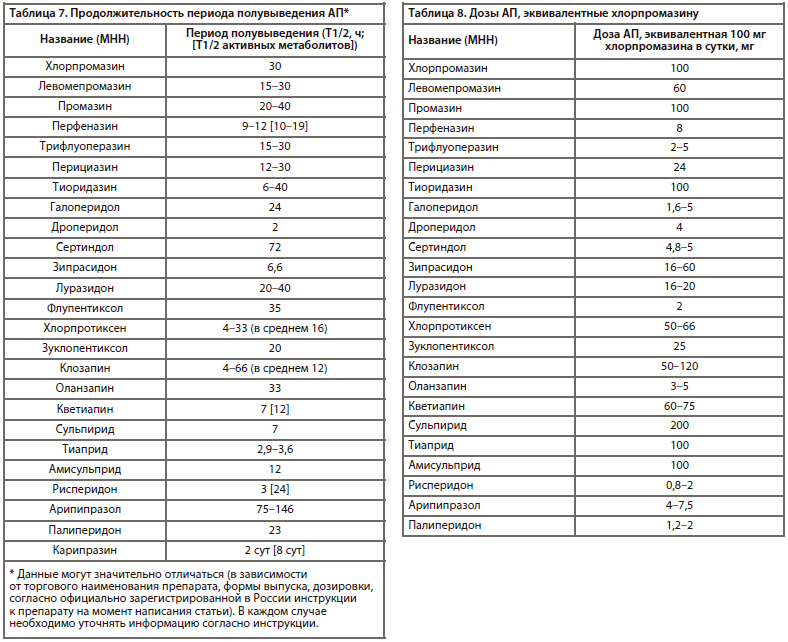
Необходимо также отметить важный фармакокинетический параметр АП — продолжительность действия (период полувыведения препарата и его активных метаболитов) [23], который представлен в табл. 7.
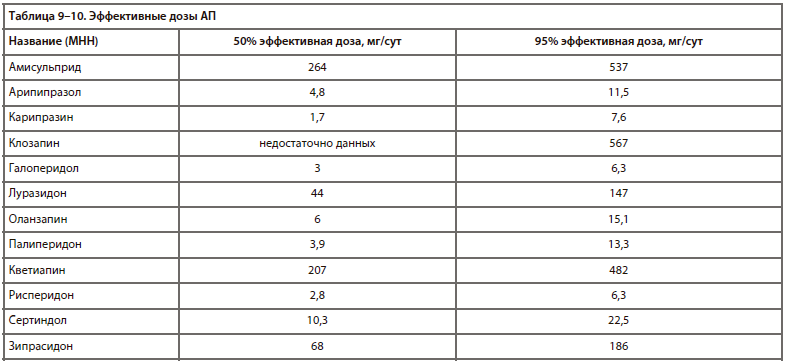
Помимо классификации по продолжительности действия, важное клиническое значение имеет сила основного (антипсихотического) действия препарата. Поэтому в этом случае оправдана концепция эквивалентности (по аналогии с условными эквивалентами анксиолитиков [34, 35] и антидепрессантов [36-38], хотя подобная практика для АП возникла раньше). Для АП выделяют хлорпромазиновый эквивалент (доза препарата, эквивалентная 100 мг хлопромазина по терапевтическому эффекту). Существуют и другие методы сравнения дозировок АП, но они менее распространены в России [40, 41, 43].
По данным ряда исследований [53, 54], эффективная суточная дозировка АП составляет от 300 до 600 мг (максимум до 900 мг\сут) в хлорпромазиновом эквиваленте, меньшие дозировки не обеспечивают достаточный отклик, а большие — не приводят к увеличению эффективности, но увеличивают риск развития побочного действия.
Указанные границы не могут представлять константу для всех случаев. Так, по данным некоторых авторов, у многих АП хлорпромазиновый эквивалент не отличается строгой линейностью (особенно при сравнении между собой типичных и атипичных АП) [39, 55], поэтому каждая клиническая ситуация требует рассмотрения инструкции к АП, клинических рекомендаций, а также других источников научной информации.
Хлорпромазиновые эквиваленты некоторых АП, по данным различных исследований [16, 39, 41, 42, 44, 45, 46, 47, 48, 49], представлены в табл. 8.
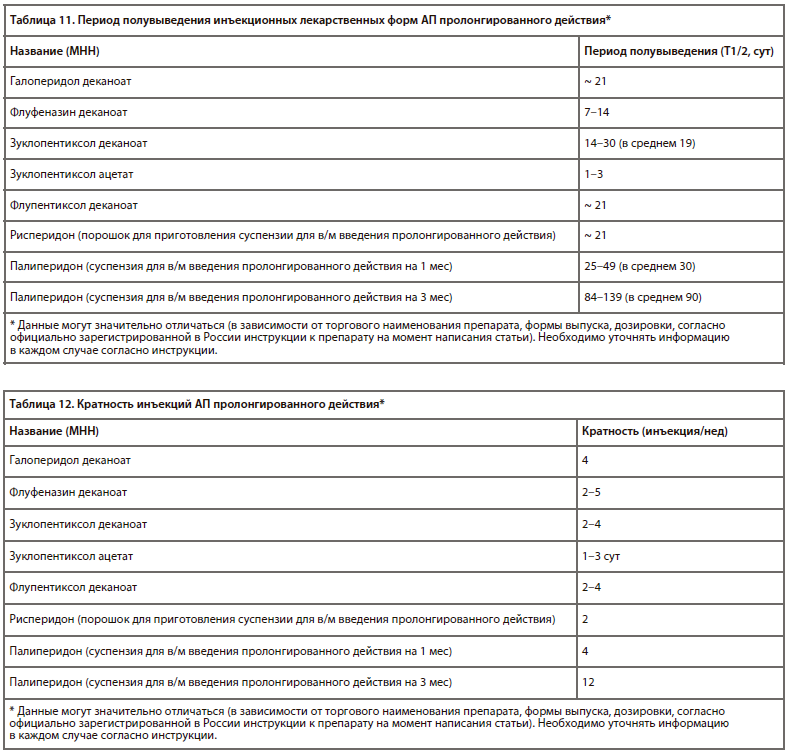
Последние актуальные исследования [50] также вводят понятие 50% и 95% эффективной дозы, т. е. дозы АП, которая у 50% и, соответственно, 95% пациентов обусловливает редукцию психотической симптоматики (табл. 9).

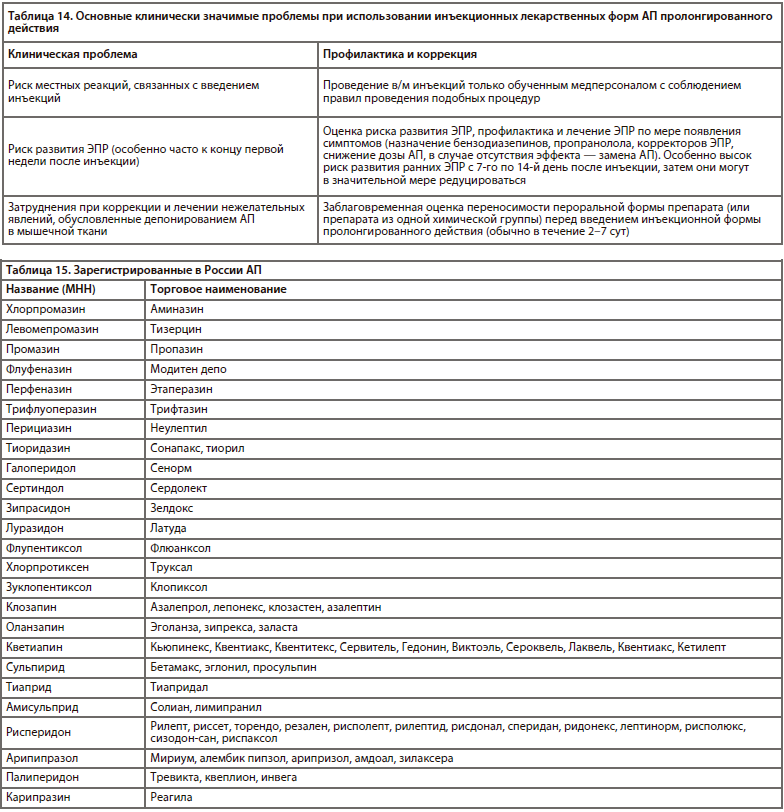
В настоящее время в России существует 8 лекарственных форм АП пролонгированного действия (5 АПП и 3 АВП): галоперидол деканоат, флуфеназин деканоат, зуклопентиксол деканоат, зуклопентиксол ацетат, флупентиксол деканоат, рисперидон (порошок для приготовления суспензии для в/м введения пролонгированного действия), палиперидон (суспензия для в/м введения пролонгированного действия на 1 мес), палиперидон (суспензия для в/м введения пролонгированного действия на 3 мес). За рубежом также существуют пролонгированные инъекционные формы арипипразола и оланзапина, которые в настоящее время в России не зарегистрированы [23].
Несмотря на большее количество недостатков применения указанных форм АП (ограниченность выбора, невозможность быстрой коррекции побочных эффектов при непереносимости, затруднения при необходимости маневра с дозировками, необходимость строгого контроля за датой последней инъекции и т.д.), в некоторых ситуациях (в случае некомплайентных пациентов) они позволяют проводить терапию более эффективно [51].
Представлены рекомендуемые дозы «пролонгов» (табл. 9–10) [23], период полувыведения основных АП (табл. 11) [23], частота инъекций (табл. 12) [23], хлорпромазиновые эквиваленты (табл. 13) [39, 44], а также основные клинически значимые проблемы, вызываемые АП (табл. 14) [51].

Антипсихотические препараты в форме раствора для приема внутрь
Растворы АП для приема внутрь в России представлены галоперидолом, рисперидоном и перициазином и применяются преимущественно для лечения детей и пожилых (учитывая удобство дозировки).
Тактика применения антипсихотических препаратов
С учетом официально зарегистрированных инструкций по применению лекарственных препаратов в настоящее время в России используют несколько тактик применения АП в клинической практике психиатрии.
Амбулаторная тактика. При острых и хронических психических расстройствах АП назначают постепенно, начиная со стартовой дозы и титруя до минимальной эффективной терапевтической дозы. Лечение проводят до наступления клинического эффекта (в среднем 1–4 нед), с возможным увеличением дозировки при неэффективности, до достижения клинической медикаментозной ремиссии и далее, от момента ее наступления, длительным курсом (не менее 1 года, в некоторых случаях до 3 и более лет, при наличии показаний), с последующим снижением дозировки до поддерживающей и рассмотрением вопроса отмены препарата (при наличии показаний).
Препараты первой линии — атипичные АП, при недостаточной эффективности — подбор терапии (замена на другой АП и лечение им в среднетерапевтических дозировках не менее 1,5–2 мес); в случае недостаточной эффективности терапии, возможно рассмотрение вопроса о переводе на терапию клозапином (при шизофрении), в случае коморбидной аффективной симптоматики возможно дополнительное назначение нормотимиков, антидепрессантов, в случае коморбидной тревожной или диссомнической симптоматики возможно (обычно кратковременное, до 1 месяца) назначение транквилизаторов.
При недостаточной приверженности к лечению (при отсутствии противопоказаний), в некоторых случаях возможно назначение поддерживающей терапии инъекционными пролонгированными формами АП (галоперидол деканоат, флуфеназин депо, зуклопентиксол депо, флупентиксол депо, пролонгированные инъекционные формы рисперидона и палиперидона); при неэффективности или недостаточной эффективности одного или нескольких курсов терапии АП — рекомендовано рассмотрение вопроса о подборе терапии в стационарных условиях.
Стационарная тактика. В целом эта тактика значительно не отличается от амбулаторной, за исключением нескольких моментов. Во-первых, увеличение дозировок не требует плавного титрования и может проводится в более быстром темпе (при ежедневном наблюдении медперсонала за пациентом); во-вторых, в случае анозогнозии (отсутствие критики к заболеванию и состоянию), а также острой симптоматики (состояние ажитации, агрессии, психомоторного возбуждения и др.) применяют первые 1–2 нед АП, в основном типичные (хлорпромазин, галоперидол, дроперидол, трифлуоперазин, левомепромазин, зуклопентиксол, тиаприд), в виде в/в и в/м инъекций. Преимущества инъекционных форм АП (за исключением препаратов пролонгированного действия) ограничены ситуациями острых состояний при отказе (до появления критики к состоянию) или невозможности их перорального приема в условиях стационара.
Существует практика назначения АП при аффективных расстройствах, нарушениях поведения, сна и в других ситуациях (например, аугментация при лечении другими группами препаратов), однако этот вопрос не является темой данной работы.
Из полного списка АП в России (по данным Государственного реестра лекарственных средств) [23] зарегистрировано не менее 27 наименований. Среди препаратов, заявленных как АП, некоторые из них фактически относятся к другим группам. Примером может служить алимемазин (по АТХ отнесен к подгруппе R06AD — производные фенотиазина группы R06A — антигистаминные препараты для системного применения) или лития карбонат (по АТХ отнесен к подгруппе N05AN группы антипсихотических препаратов, а фактически является нормотимиком).
Зарегистрированные в России в настоящее время АП с учетом торговых наименований представлены в табл. 15 (за исключением торговых наименований формы «МНН», «МНН — фирма-производитель» или «МНН — лекарственная форма») [23, 29].
Помимо длительности действия (период полувыведения), клинического эффекта, важно учитывать при назначении препарата его цену и доступность (законодательное регулирование льготного лекарственного обеспечения, политика аптечных сетей). В силу этого окончательное решение о назначении того или иного препарата лечащий врач принимает с учетом показаний, противопоказаний, клинического профиля препартата, его фармакодинамики, фармакокинетики, индивидуальных особенностей пациента и указанных выше параметров.
Важные аспекты, которые необходимо учитывать при назначении АП, включают также возраст, влияние на управление транспортными средствами и деятельность, требующую повышенной концентрации внимания, а также период беременности и лактации (согласно официальным инструкциям к лекарственным средствам) [23, 29] (табл. 16).
Сведения об авторе:
Маслов Константин Андреевич — врач-психиатр ГБУЗ ОПБ им. К.Р. Евграфова (г. Пенза). E-mail:skrembler@mail.ru
Психиатрия Психиатрия и психофармакотерапия им. П.Б. Ганнушкина
№05 2020
Антипсихотические препараты в современной клинической практике врача-психиатра в России. Вопросы прикладного применения №05 2020
Номера страниц в выпуске:41-50
Резюме
Открытие первого антипсихотического препарата — хлорпромазина — состоялось в 1950 г., и с тех пор список антипсихотиков значительно расширился. Эти препараты с успехом используются при психотических расстройствах и некоторых других нарушениях. В статье не только рассмотрены не только основные области применения антипсихотических препаратов, но и освещены основные клинически значимые проблемы при их применении, а также меры профилактики и коррекции побочных эффектов.
Ключевые слова: антипсихотики, нейролептики, антипсихотики в России, нейролептики в России.
Для цитирования: К.А. Маслов. Антипсихотические препараты в современной клинической практике врача-психиатра в России. Вопросы прикладного применения. Психиатрия и психофармакотерапия. 2020; 5: 41–50.
Открытие первого антипсихотического препарата — хлорпромазина — состоялось в 1950 г., и с тех пор список антипсихотиков значительно расширился. Эти препараты с успехом используются при психотических расстройствах и некоторых других нарушениях. В статье не только рассмотрены не только основные области применения антипсихотических препаратов, но и освещены основные клинически значимые проблемы при их применении, а также меры профилактики и коррекции побочных эффектов.
Ключевые слова: антипсихотики, нейролептики, антипсихотики в России, нейролептики в России.
Для цитирования: К.А. Маслов. Антипсихотические препараты в современной клинической практике врача-психиатра в России. Вопросы прикладного применения. Психиатрия и психофармакотерапия. 2020; 5: 41–50.
Antipsychotics in Russia, in modern clinical practice of the psychiatrist. Questions of applied practical application
K.A. MaslovK.R. Evgrafov Regional mental hospital 440026, Russian Federation, Penza, ul. Lermontova, d. 28. skrembler@mail.ru
Abstract
The discovery of the first antipsychotic drug - chlorpromazine - took place in 1950 and since then the list of antipsychotics has expanded significantly. These drugs are used successfully for psychotic disorders and some other disorders. The article discusses not only the main areas of application of antipsychotic drugs, but also highlights the main clinically significant problems in their use, as well as the prevention and correction of side effects.
Keywords: antipsychotics, antipsychotics in Russia, neuroleptics, neuroleptics in Russia.
For citation: K.A. Maslov. Antipsychotics in Russia, in modern clinical practice of the psychiatrist. Questions of applied practical application. Psychiatry and psychopharmacotherapy. 2020; 5: 41–50.
Введение
Антипсихотические препараты (далее антипсихотики, АП) — препараты, применяемые, прежде всего, при психотических расстройствах и (реже) других нарушениях, связанных с обменом нейромедиаторов (преимущественно дофамина) в области головного мозга (аффективные и поведенческие нарушения, тревожные расстройства, нарушения сна и др.).Согласно международной анатомо-терапевтическо-химической классификации (далее АТХ), антипсихотические препараты относятся к группе препаратов для лечения заболеваний нервной системы N5 (психолептики), подгруппе N05A (антипсихотические препараты) [1].
Ранее (с конца 1950-х годов) при обозначении АП применяли термины: «нейролептики» [2, 3], «нейроплегики», «большие транквилизаторы» (по аналогии с «малыми транквилизаторами»), «атарактики» [2]. В настоящее время в России используют термин «антипсихотики первого поколения» (далее АПП), «типичные нейролептики», «типики», «классические нейролептики»; за рубежом — first generation antipsychotics или FGA. Существуют также препараты, называемые в России антипсихотиками второго поколения (далее АВП), «атипичные нейролептики», «атипики», или «атипичные антипсихотики») [3], а за рубежом — atypical antipsychotics (AAP), second generation antipsychotics (SGA). По мнению ряда исследователей, основные различия между ААП и АВП заключаются в разном рецепторном профиле и, как следствие, в разной степени представленности и выраженности лечебных и побочных эффектов (прежде всего по выраженности экстрапирамидных расстройств) [4].
Краткая история антипсихотических препаратов
До открытия АП (в 1950-е годы) при психотических расстройствах применяли барбитураты, внутривенные вливания раствора кальция, препараты брома и другие, более экзотические, с точки зрения современной медицины, способы (подкожные вдувания кислорода, введение лошадиной сыворотки, сыворотки от других пациентов, находящихся в состоянии ремиссии, и др.) [5]. Значительное распространение получили также инсулинокоматозная [6], электросудорожная терапия [7] и психохирургия (лоботомия) [8].Первый АП — хлорпромазин — был открыт в 1950 г. [9] в процессе синтеза антигистаминных препаратов [10]. Этот препарат сразу и надолго стал «золотым стандартом» и эталоном среди АП. Затем были синтезирован ряд других АП, среди которых наибольшее применение получил галоперидол (в клинической практике с 1959 г.) [11].
В табл. 1 отражено время широкого внедрения некоторых АП в клиническую практику [10-19].

Лечебные и побочные эффекты
Механизм действия АП обусловлен, преимущественно, блокадой дофаминовых (в первую очередь D2-типа) рецепторов в дофаминергических путях (главным образом в мезолимбическом) и в меньшей степени, серотониновых (5-HT2A) и других рецепторов (α-адренорецепторов, м-холинорецепторов, H1-гистаминорецепторов и др.) в центральной и периферической нервной системе. Исходя из этого, применение АП вызывает:• антипсихотический эффект (основной);
• седативный эффект;
• нормотимический эффект.
Системное воздействие препарата влечет за собой и ряд побочных эффектов, которые могут вызывать ряд осложнений. В первую очередь, они обусловлены дофаминергической блокадой в мезокортикальном, нигростриальном и тубероинфундибулярном путях, а также влиянием на различные рецепторы в периферических отделах нервной системы. Наиболее часто в числе осложнений встречаются нейролептические экстрапирамидные расстройства (ЭПР), метаболические нарушения (ожирение, диабет, гиперпролактинемия), избыточная седация, вторичная дефицитарная симптоматика, слабость, когнитивные нарушения, нейролептическая депрессия, психозы отмены, сексуальные нарушения, слюнотечение. Отмечается также кардио- и гепатотоксичность АП.
Соотношение выраженности лечебных и побочных эффектов между собой определяет спектр клинического действия препарата.
Для большинства АП (кроме, вероятно, клозапина и некоторых современных атипичных АП) развитие антипсихотического эффекта происходит при блокаде, приблизительно, 60–65% дофаминовых рецепторов D2-типа. При блокаде свыше 70–80% рецепторов увеличивается риск развития ЭПР [20-2]. В соответствии с этим условно выделяют «терапевтическое окно» дозировок, в котором АП вызывают собственно антипсихотический эффект. Рекомендуемый диапазон доз АП в России (стартовые и максимальные суточные) [23] представлен в табл. 2.
Основные клинически значимые проблемы применения АП и методы их профилактики и коррекции представлены в табл. 3.
В настоящее время в России (по некоторым оценкам) [29-31] наиболее популярны из группы АП в клинической практике (в порядке убывания): рисперидон, сульпирид, тиоридазин, кветиапин.
Для сравнения — в США (по некоторым оценкам) [32] из группы АП наиболее популярны в клинической практике (в порядке убывания): кветиапин, арипипразол, рисперидон.

Классификация антипсихотических препаратов
Существует несколько классификаций АП, которые опираются на их поколение, механизм действия, химическое строение и другие критерии.Зарегистрированные в настоящее время в России АП могут подразделяться по их механизму действия, согласно АТХ (табл. 4) [1].
АП также подразделяют в зависимости от поколения на типичные и атипичные (по механизму действия, представленности и выраженности побочных и лечебных эффектов) [4] (табл. 5).
Помимо перечисленных классификаций в отечественной психиатрии существовало, множество различных эклектичных классификаций АП по различным неспецифичным признакам, однако в настоящее время они устарели.
Одна из наиболее прогрессивных и логичных классификаций АП основана на рецепторном механизме действия [26]. Такой подход логичен, последователен и отлично дополняет существующие классификации (табл. 6).
Необходимо также отметить важный фармакокинетический параметр АП — продолжительность действия (период полувыведения препарата и его активных метаболитов) [23], который представлен в табл. 7.
Помимо классификации по продолжительности действия, важное клиническое значение имеет сила основного (антипсихотического) действия препарата. Поэтому в этом случае оправдана концепция эквивалентности (по аналогии с условными эквивалентами анксиолитиков [34, 35] и антидепрессантов [36-38], хотя подобная практика для АП возникла раньше). Для АП выделяют хлорпромазиновый эквивалент (доза препарата, эквивалентная 100 мг хлопромазина по терапевтическому эффекту). Существуют и другие методы сравнения дозировок АП, но они менее распространены в России [40, 41, 43].
По данным ряда исследований [53, 54], эффективная суточная дозировка АП составляет от 300 до 600 мг (максимум до 900 мг\сут) в хлорпромазиновом эквиваленте, меньшие дозировки не обеспечивают достаточный отклик, а большие — не приводят к увеличению эффективности, но увеличивают риск развития побочного действия.
Указанные границы не могут представлять константу для всех случаев. Так, по данным некоторых авторов, у многих АП хлорпромазиновый эквивалент не отличается строгой линейностью (особенно при сравнении между собой типичных и атипичных АП) [39, 55], поэтому каждая клиническая ситуация требует рассмотрения инструкции к АП, клинических рекомендаций, а также других источников научной информации.
Хлорпромазиновые эквиваленты некоторых АП, по данным различных исследований [16, 39, 41, 42, 44, 45, 46, 47, 48, 49], представлены в табл. 8.
Последние актуальные исследования [50] также вводят понятие 50% и 95% эффективной дозы, т. е. дозы АП, которая у 50% и, соответственно, 95% пациентов обусловливает редукцию психотической симптоматики (табл. 9).





Инъекционные лекарственные формы антипсихотических препаратов пролонгированного действия
Необходимо отдельно остановиться на инъекционных лекарственных формах АП пролонгированного действия («пролонги»), которые применяют для длительного поддерживающего лечения и профилактики обострений шизофрении (преимущественно) и некоторых других психических расстройств (в подавляющем большинстве психотического уровня). Кратность инъекций может колебаться от нескольких дней до 3 мес (в среднем 2–4 нед), что достигается путем растворения сложных эфиров АП в растворе растительного масла (кунжутное, кокосовое) и другими способами (микрогранулы пролонгированного высвобождения и др.).В настоящее время в России существует 8 лекарственных форм АП пролонгированного действия (5 АПП и 3 АВП): галоперидол деканоат, флуфеназин деканоат, зуклопентиксол деканоат, зуклопентиксол ацетат, флупентиксол деканоат, рисперидон (порошок для приготовления суспензии для в/м введения пролонгированного действия), палиперидон (суспензия для в/м введения пролонгированного действия на 1 мес), палиперидон (суспензия для в/м введения пролонгированного действия на 3 мес). За рубежом также существуют пролонгированные инъекционные формы арипипразола и оланзапина, которые в настоящее время в России не зарегистрированы [23].
Несмотря на большее количество недостатков применения указанных форм АП (ограниченность выбора, невозможность быстрой коррекции побочных эффектов при непереносимости, затруднения при необходимости маневра с дозировками, необходимость строгого контроля за датой последней инъекции и т.д.), в некоторых ситуациях (в случае некомплайентных пациентов) они позволяют проводить терапию более эффективно [51].
Представлены рекомендуемые дозы «пролонгов» (табл. 9–10) [23], период полувыведения основных АП (табл. 11) [23], частота инъекций (табл. 12) [23], хлорпромазиновые эквиваленты (табл. 13) [39, 44], а также основные клинически значимые проблемы, вызываемые АП (табл. 14) [51].






Антипсихотические препараты в форме раствора для приема внутрь
Растворы АП для приема внутрь в России представлены галоперидолом, рисперидоном и перициазином и применяются преимущественно для лечения детей и пожилых (учитывая удобство дозировки).
Тактика применения антипсихотических препаратов
С учетом официально зарегистрированных инструкций по применению лекарственных препаратов в настоящее время в России используют несколько тактик применения АП в клинической практике психиатрии.
Амбулаторная тактика. При острых и хронических психических расстройствах АП назначают постепенно, начиная со стартовой дозы и титруя до минимальной эффективной терапевтической дозы. Лечение проводят до наступления клинического эффекта (в среднем 1–4 нед), с возможным увеличением дозировки при неэффективности, до достижения клинической медикаментозной ремиссии и далее, от момента ее наступления, длительным курсом (не менее 1 года, в некоторых случаях до 3 и более лет, при наличии показаний), с последующим снижением дозировки до поддерживающей и рассмотрением вопроса отмены препарата (при наличии показаний).
Препараты первой линии — атипичные АП, при недостаточной эффективности — подбор терапии (замена на другой АП и лечение им в среднетерапевтических дозировках не менее 1,5–2 мес); в случае недостаточной эффективности терапии, возможно рассмотрение вопроса о переводе на терапию клозапином (при шизофрении), в случае коморбидной аффективной симптоматики возможно дополнительное назначение нормотимиков, антидепрессантов, в случае коморбидной тревожной или диссомнической симптоматики возможно (обычно кратковременное, до 1 месяца) назначение транквилизаторов.
При недостаточной приверженности к лечению (при отсутствии противопоказаний), в некоторых случаях возможно назначение поддерживающей терапии инъекционными пролонгированными формами АП (галоперидол деканоат, флуфеназин депо, зуклопентиксол депо, флупентиксол депо, пролонгированные инъекционные формы рисперидона и палиперидона); при неэффективности или недостаточной эффективности одного или нескольких курсов терапии АП — рекомендовано рассмотрение вопроса о подборе терапии в стационарных условиях.
Стационарная тактика. В целом эта тактика значительно не отличается от амбулаторной, за исключением нескольких моментов. Во-первых, увеличение дозировок не требует плавного титрования и может проводится в более быстром темпе (при ежедневном наблюдении медперсонала за пациентом); во-вторых, в случае анозогнозии (отсутствие критики к заболеванию и состоянию), а также острой симптоматики (состояние ажитации, агрессии, психомоторного возбуждения и др.) применяют первые 1–2 нед АП, в основном типичные (хлорпромазин, галоперидол, дроперидол, трифлуоперазин, левомепромазин, зуклопентиксол, тиаприд), в виде в/в и в/м инъекций. Преимущества инъекционных форм АП (за исключением препаратов пролонгированного действия) ограничены ситуациями острых состояний при отказе (до появления критики к состоянию) или невозможности их перорального приема в условиях стационара.
Существует практика назначения АП при аффективных расстройствах, нарушениях поведения, сна и в других ситуациях (например, аугментация при лечении другими группами препаратов), однако этот вопрос не является темой данной работы.
Из полного списка АП в России (по данным Государственного реестра лекарственных средств) [23] зарегистрировано не менее 27 наименований. Среди препаратов, заявленных как АП, некоторые из них фактически относятся к другим группам. Примером может служить алимемазин (по АТХ отнесен к подгруппе R06AD — производные фенотиазина группы R06A — антигистаминные препараты для системного применения) или лития карбонат (по АТХ отнесен к подгруппе N05AN группы антипсихотических препаратов, а фактически является нормотимиком).
Зарегистрированные в России в настоящее время АП с учетом торговых наименований представлены в табл. 15 (за исключением торговых наименований формы «МНН», «МНН — фирма-производитель» или «МНН — лекарственная форма») [23, 29].
Помимо длительности действия (период полувыведения), клинического эффекта, важно учитывать при назначении препарата его цену и доступность (законодательное регулирование льготного лекарственного обеспечения, политика аптечных сетей). В силу этого окончательное решение о назначении того или иного препарата лечащий врач принимает с учетом показаний, противопоказаний, клинического профиля препартата, его фармакодинамики, фармакокинетики, индивидуальных особенностей пациента и указанных выше параметров.
Важные аспекты, которые необходимо учитывать при назначении АП, включают также возраст, влияние на управление транспортными средствами и деятельность, требующую повышенной концентрации внимания, а также период беременности и лактации (согласно официальным инструкциям к лекарственным средствам) [23, 29] (табл. 16).
Заключение
АП — одна из групп препаратов (наряду с антидепрессантами и анксиолитиками), появление которой со второй половины двадцатого века ознаменовало начало «психофармакологической эры» в истории психиатрии. Возможности этих препаратов кардинальным образом изменили облик психиатрии и подход к лечению пациентов с психическими расстройствами. Механизм действия АП, побочные эффекты, особенности применения и сопутствующие проблемы, а также пути их преодоления хорошо изучены в многочисленных научных исследованиях, многие из них внесены в «Примерный перечень ВОЗ основных лекарственных средств» [52]. В настоящее время АП представляют основу длительной патогенетической терапии психотических расстройств и других коморбидных им состояний и расстройств.Сведения об авторе:
Маслов Константин Андреевич — врач-психиатр ГБУЗ ОПБ им. К.Р. Евграфова (г. Пенза). E-mail:skrembler@mail.ru
Список исп. литературыСкрыть список1. WHO Collaborating Centre for Drug Statistics Methodology [Electronic resource]. URL: http://www.whocc.no. (access date: 02.04.2020).
2. King C, Voruganti LN. What's in a name? The evolution of the nomenclature of antipsychotic drugs. Journal of Psychiatry & Neuroscience. 2002; May. 27(3): 168–75.
3. Мосолов С.Н. Современная антипсихотическая фармакотерапия шизофрении. Неврология. Психиатрия. 2004; 10. / Mosolov S.N. Sovremennaya antipsixoticheskaya farmakoterapiya shizofrenii. Nevrologiya. Psixiatriya. 2004; 10. [in Russian].
4. A Roadmap to Key Pharmacologic Principles in Using Antipsychotics. The Primary Care Companion to the Journal of Clinical Psychiatry. 2007; 9 (6): 444–54.
5. Гиляровский В.А. Психиатрия. 2 изд. Москва-Ленинград, БИОМЕДГИЗ, 1935 / Gilyarovskij V.A. Psixiatriya. 2 izd. Moskva-Leningrad, BIOMEDGIZ, 1935. [in Russian].
6. Jones K. Insulin coma therapy in schizophrenia. J Roy Soc Med. 2000; Mar. 93 (3): 147–149.
7. Cerletti, U. Electroshock therapy. In AM Sackler et al. (eds). The Great Physiodynamic Therapies in Psychiatry: an historical appraisal. New York: Hoeber-Harper, 1956. 91–120.
8. Levinson H. The strange and curious history of lobotomy. BBC News. [Electronic resource]. 8 November 2011. URL: http://www.bbc.com/news/magazine-15629160 (access date: 02.04.2020).
9. López-Muñoz F, Alamo C, Cuenca E, et al. History of the discovery and clinical introduction of chlorpromazine. Annals of Clinical Psychiatry. 2005; Jul-Sep. 17 (3): 113–35.
10. Healy D. Explorations in a new world. The creation of psychopharmacology. Harvard University Press. 2004; 77.
11. Granger B, Albu S. The haloperidol story. Ann Clin Psychiatry. 2005; 17(3): 137–140.
12. Riederer P, Laux G, Pöldinger W. Neuro-Psychopharmaka: Ein Therapie-Handbuch Band 1: Allgemeine Grundlagen der Pharmakopsychiatrie. Springer-Verlag Wien GmbH: 1992; 63.
13. Walter S. Drug discovery: a history. New York: Wiley. 2005; 410.
14. Andrew W. Pharmaceutical Manufacturing Encyclopedia. Elsevier. 2013; 1102.
15. Taylor D, Paton C, Kapur S. The Maudsley Prescribing Guidelines in Psychiatry (12th ed.). London, U K: Wiley-Blackwell. 2015.
16. Riedel M, Müller N, Strassnig M et al. Quetiapine in the treatment of schizophrenia and related disorders. Neuropsychiatric Disease and Treatment. 2007; Apr. 3(2): 219–235.
17. Bachmann C, Lempp T, Glaeske G et al. Antipsychotic prescriptions in children and adolescents—an analysis of data from a German statutory health insurance company from 2005–2012. Dtsch Arztebl Int 2014; 111(3): 25–34.
18. U.S. Food and Drug Administration Center for Drug Evaluation and Research. Novel Drug Approvals for 2015. URL: http://www.fda.gov/drugs/ new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2015 (access date: 02.04.2020).
19. Product information for AusPAR Latuda Commercial Eyes Pty Ltd. Product information Latuda (lurasidone hydrochloride) (PDF). 20 June 2014; URL: http://www.tga.gov.au/sites/default/files/auspar-lurasidone-hydrochloride-140620-pi.pdf (access date: 02.04.2020).
20. Farah A. Atypicality of atypical antipsychotics. Prim Care Companion J Clin Psychiatry. 2005. 7(6): 268–74.
21. Kapur S, Zipursky R, Jones C, et al. Relationship Between Dopamine D(2) Occupancy, Clinical Response, and Side Effects: A Double-Blind PET Study of First-Episode Schizophrenia. Am J Psychiatry. 2000; Apr. 157(4): 514–20.
22. Kapur S, Zipursky R, Remington G. Clinical and Theoretical Implications of 5-HT2 and D2 Receptor Occupancy of Clozapine, Risperidone, and Olanzapine in Schizophrenia. Am J Psychiatry. 1999; Feb. 156(2): 286–93.
23. Государственный реестр лекарственных средств [Электронный ресурс]. URL: http://grls.rosminzdrav.ru. (Дата обращения: 02.04.2020). / Gosudarstvennyj reestr lekarstvennyh sredstv [Elektronnyj resurs]. URL: http://grls.rosminzdrav.ru. (Data obrashcheniya: 02.04.2020). [in Russian].
24. Дробижев М.Ю. Кардиологические аспекты проблемы переносимости и безопасности нейролептика. Психиатрия и психофармакотерапия. 2004; 6(2). / Drobizhev M.Yu. Kardiologicheskie aspekty` problemy` perenosimosti i bezopasnosti nejroleptika. Psixiatriya i psixofarmakoterapiya. 2004; 6(2). [in Russian].
25. Жаркова Н.Б. Переход к терапии атипичными антипсихотическими средствами — вопрос стратегии или тактики психофармакотерапии? Психиатрия и психофармакотерапия. 2001; 3(1). / Zharkova N.B. Perexod k terapii atipichny`mi antipsixoticheskimi sredstvami — vopros strategii ili taktiki psixofarmakoterapii? Psixiatriya i psixofarmakoterapiya. 2001; 3(1). [in Russian].
26. Дробижев М.Ю., Овчинников А.А. Зачем нужна современная классификация антипсихотиков? Русский медицинский журнал. 2012; 20(29): 1449–1457. / Drobizhev M.YU., Ovchinnikov A.A. Zachem nuzhna sovremennaya klassifikaciya antipsihotikov? Russkij medicinskij zhurnal. 2012; 20(29): 1449–1457. [in Russian].
27. Марценковский И.А., Марценковская И.И. Эпилепсия и коморбидные расстройства психики и поведения. Український медичний часопис. 2015; 4: 43–53. / Marcenkovskij I.A., Marcenkovskaya I.I. E`pilepsiya i komorbidny`e rasstrojstva psixiki i povedeniya. Ukraїns`kij medichnij chasopis. 2015; 4: 43–53. [in Russian].
28. Masaomi I, Shigenori T, Nobuhisa K et al. Optimal Extent of Dopamine D2 Receptor Occupancy by Antipsychotics for Treatment of Dopamine Supersensitivity Psychosis and Late-Onset Psychosis. J Clin Psychopharmacol. 2013; Jun. 33(3): 398–404.
29. Регистр лекарственных средств России [Электронный ресурс]. URL: http://www.rlsnet.ru (Дата обращения: 02.04.2020). / Registr lekarstvennyh sredstv Rossii [Elektronnyj resurs]. URL: http://www.rlsnet.ru (Data obrashcheniya: 02.04.2020). [in Russian].
30. Рациональный выбор лекарств на основе индекса информационного спроса [Электронный ресурс]. Газета РЛС. 2008. №3. URL: http://www.rlsnet.ru/news_295.htm. (Дата обращения: 02.04.2020). / Racional'nyj vybor lekarstv na osnove indeksa informacionnogo sprosa [Elektronnyj resurs]. Gazeta RLS. 2008. №3. URL: http://www.rlsnet.ru/ news_295.htm. (Data obrashcheniya: 02.04.2020). [in Russian].
31. Вышковский Г.Л. Методология оптимального выбора фаз маркетингового воздействия при медиапланировании. Реклама. Теория и практика. 2007; 02(20). / Vyshkovskij G.L. Metodologiya optimal'nogo vybora faz marketingovogo vozdejstviya pri mediaplanirovanii. Reklama. Teoriya i praktika. 2007; 02(20). [in Russian].
32. Free U.S. Outpatient Drug Usage Statistics [Electronic resource]. ClinCalc DrugStats Database. URL: http://clincalc.com/DrugStats/ (access date: 02.04.2020).
33. Справочное руководство по психофармакологическим и противоэпилептическим препаратам, разрешенным к применению в России. Под ред. С.Н. Мосолова. — Изд. 2-е, перераб. — М.: Издательство БИНОМ, 2004; 10–11, 43. / Spravochnoe rukovodstvo po psixofarmakologicheskim i protivoe`pilepticheskim preparatam, razreshenny`m k primeneniyu v Rossii. Pod red. S. N. Mosolova. — Izd. 2-e, pererab. — M.: Izdatel`stvo BINOM, 2004; 10–11, 43. [in Russian].
34. Ashton H. Drug newsletter. Northern Regional Health Authority. Newcastle upon Tyne: Wolfson Unit of Clinical Pharmacology Royal Victoria Infirmary [Electronic resource]. 1985; Apr 31. URL: http://benzo.org.uk/drcha.htm (access date: 02.04.2020).
35. Benzodiazepine Equivalency Table [Electronic resource]. 2007; Revised April. URL: http://benzo.org.uk/bzequiv.htm. (access date: 02.04.2020).
36. Jakubovski E, Varigonda AL, Freemantle N et al. Systematic Review and Meta-Analysis: Dose-Response Relationship of Selective Serotonin Reuptake Inhibitors in Major Depressive Disorder. The American Journal of Psychiatry. 2016; Feb 1; 173 (2): 174–83.
37. Hayasaka Y, Purgato M, Magni LR, et al. Dose equivalents of antidepressants: Evidence-based recommendations from randomized controlled trials. J Affect Disord. 2015; 180: 179-184. doi:10.1016/j.jad.2015.03.021
38. Ali IM. Long-term treatment with antidepressants in primary care: Are sub-therapeutic doses still being used? Psychiatric Bulletin. 1998; Jan; 22 (1): 15-19.
39. Atkins M, Burgess A, Bottomley C et al. Chlorpromazine equivalents: a consensus of opinion for both clinical and research applications. Psychiatric Bulletin. 1997; Apr. 21(4): 224–226.
40. Leucht S, Samara M, Heres S et al. Dose Equivalents for Antipsychotic Drugs: The DDD Method. Schizophrenia Bulletin. 2016; 42 Suppl 1(Suppl 1): S90–4.
41. Andreasen N, Pressler M, Nopoulos P et al. Antipsychotic Dose Equivalents and Dose-Years: A Standardized Method for Comparing Exposure to Different Drugs. Biol Psychiatry. 2010; 67(3): 255–262.
42. Woods S. Chlorpromazine Equivalent Doses for the Newer Atypical Antipsychotics. J Clin Psychiatry. 2003; 64(6): 663–7.
43. Rijcken C, Monster T, Brouwers J et al. Chlorpromazine Equivalents Versus Defined Daily Doses: How to Compare Antipsychotic Drug Doses? J Clin Psychopharmacol. 2003; 23(6): 657–9.
44. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: Comparing apples and oranges! Indian J Psychiatry. 2013; 55(2): 207–208.
45. Leucht S, Samara M, Heres S et al. Dose equivalents for second-generation antipsychotics: the minimum effective dose method. Schizophrenia Bulletin. 2014; 40 (2): 314–326.
46. Leucht S, Samara M, Heres S et al. Dose equivalents for second-generation antipsychotic drugs: the classical mean dose method. Schizophr Bull. 2015; 41(6): 1397–402.
47. Patel M, Arista I, Taylor M et al. How to compare doses of different antipsychotics: a systematic review of methods. Schizophr Res. 2013; 149(1–3): 141–8.
48. Schatzberg A, Cole J, DeBattista C. Manual of Clinical Psychopharmacology. American Psychiatric Publishing. 7th Edition. 2010.
49. Федеральное руководство по использованию лекарственных средств (формулярная система). Под ред. А.Г. Чучалина, Ю.Б. Белоусова, С.П. Голицина и др. Выпуск XIV. М.: Человек и лекарство, 2013. / Federal'noe rukovodstvo po ispol'zovaniiu lekarstvennykh sredstv formuliarnaia sistema). Pod red. A.G.Chuchalina, Iu.B.Belousova, S.P.Golitsina i dr. Vypusk XIV. M.: Chelovek i lekarstvo, 2013. [in Russian].
50. Leucht S, Crippa A, Siafis S et al. Dose-Response Meta-Analysis of Antipsychotic Drugs for Acute Schizophrenia. American Journal of Psychiatry. 2020; 177(4): 342–353.
51. Пролонгированные нейролептики: между прошлым и будущим. В центре внимания. НЦПЗ РАМН. [Электронный ресурс]. URL: http://www.psychiatry.ru/stat/112. (Дата обращения: 02.04.2020). / Prolongirovanny`e nejroleptiki: mezhdu proshly`m i budushhim. V centre vnimaniya. NCzPZ RAMN. [Elektronnyj resurs]. URL: http://www.psychiatry.ru/stat/112. (Data obrashheniya: 02.04.2020). [in Russian].
52. WHO Model List of Essential Medicines 18th list (April 2013) (Final Amendments — October 2013) [Electronic resource]. URL: http://www.who.int/iris/bitstream/10665/93142/1/EML_18_eng.pdf. (access date: 02.04.2020).
53. Gardner D, Murphy A, O’Donnell H. et al. International consensus study of antipsychotic dosing. Am J Psychiatry. 2010; 167(6): 686–93.
54. Sweileh W, Odeh J, Shraim N et al. Evaluation of Defined Daily Dose, percentage of British National Formulary maximum and chlorpromazine equivalents in antipsychotic drug utilization. Saudi Pharmaceutical Journal. 2014; 22(2): 127–32.
55. Филилеева О.В., Мосолов С.Н. Эквивалентные дозировки антипсихотических препаратов. Современная терапия психических расстройств. 2019; 3: 36–44. / Filileeva O.V., Mosolov S.N. E`kvivalentny`e dozirovki antipsixoticheskix preparatov. Sovremennaya terapiya psixicheskix rasstrojstv. 2019; 3: 36–44. [in Russian].
